What is the Difference Between nButane and Butane
5 (364) · € 14.00 · En Stock
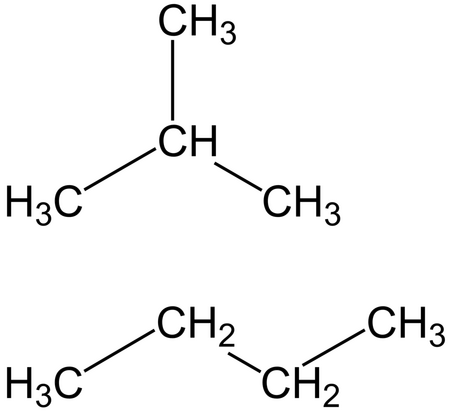
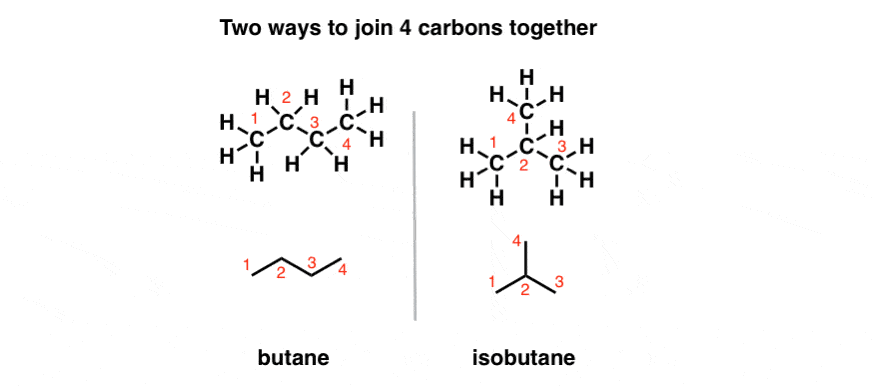
By Evan Dix July 19, 2020 Butane (C4H10) is a four carbon molecule connected by single action bonds. However, there are isomers of butane that have the same formula (C4H10) but a different geometrical arrangement. Isobutane is an example of this. Isobutane also has the formula C4H10 but it is a different molecule all together. Isobutane can be represented as i-butane and normal butane can be represented as n-butane. The "n" in the front is letting you know that you are talking about normal buta

Isomers of Butane Structural Isomers of Butane - Definition, Explanation

what is the difference between n butane,iso butane and neo butane??

Butane vs Butene: Common Misconceptions and Accurate Usage
How do butene and butane differ? - Quora

The Difference Between Propane And Butane

What are isomers ? Write the isomers of butane and pentane.

Table 1 from n-Butane and Isobutane CAS Registry Number : n-Butane : 106-978 Isobutane : 75-28-5

Org Chem Text:Chapter 1:sec1-7:1-7
Butane, Overview, Formula & Structure - Lesson

Q27- what's the difference between isobutane and sec butane 27 What is the common name for the - Chemistry - Haloalkanes and Haloarenes - 13747573
Don't Be Futyl, Learn The Butyls - Master Organic Chemistry












